
Current Litigation

Video Game Addiction Lawsuit
Video games made by Roblox Corp. (Roblox), Epic Games (Fortnite), and Microsoft/Mojang Studios (Minecraft) have grown in popularity over the past several years—especially among children. Studies have shown these games are addicting to minor children, however, leading to mental health disorders including eating disorders, anxiety, depression and suicidal ideation.
Depo-Provera Lawsuit
Women across the country are suing manufacturers of Depo-Provera (medroxyprogesterone acetate) upon receiving devastating diagnoses of brain or spinal tumors known as meningiomas. These women had been injected with Depo-Provera, a hormonal birth control taken as a shot every three months. Plaintiffs claim the drug makers were aware of Depo-Provera’s risks, but they were never informed.
CooperSurgical Embryo Solution Lawsuit

Bard PowerPort Lawsuit

Tenofovir (TDF) HIV Drug Lawsuit

Paraquat Lawsuit

Baby Powder (Talc) Lawsuit
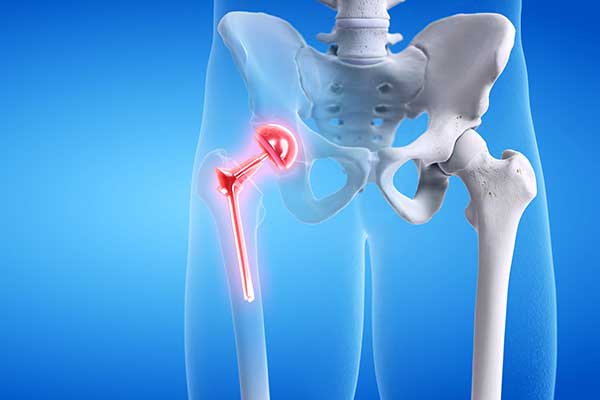
Hip Implant Lawsuit
Discuss Your Potential Claim With Our Attorneys Today
If you have suffered an injury due to a defective drug, medical device, or an environmental catastrophe, please contact us at (888) 984-7988 to discuss your potential case with a G&E attorney.
